Effect of Different Concentrations of Carbon Dioxide on Physicochemical Traits of Grapes During Storage
Muhammad Imlak, Muhammad Atif Randhawa, Ali Hassan, Naveed Ahmad and Sidrah Rafique
Muhammad Imlak*, Muhammad Atif Randhawa, Ali Hassan, Naveed Ahmad and Sidrah Rafique
Food Safety and Quality Management, University of Agriculture Faisalabad, Pakistan
- *Corresponding Author:
- Muhammad Imlak
Food Safety and Quality Management, University of Agriculture Faisalabad, Pakistan.
E-mail: imlak.khalid@yahoo.com
Received date: February 14, 2017; Accepted date: February 17, 2017; Published date: February 28, 2017
Citation: Imlak M, Randhawa MA, Hassan A, et al. Effect of Different Concentrations of Carbon Dioxide on Physicochemical Traits of Grapes During Storage. J Nutraceuticals Food Sci. 2017, 2:1
Abstract
Grapes are consumed globally as table fruit and in wine making but due to its delicate nature and soft texture, a significant portion of this valuable fruit is lost after harvest. Major factor behind its postharvest decay is fungal pathogen, Botrytis cinerea. Use of synthetic fungicides is a traditional approach to suppress this pathogen but after various health issues of fungicide residues being reported, this study was conducted as safe integrated approach involving tap water washing, CaCl2 dipping and modified atmosphere storage. Grapes were divided in 4 equal lots, 1st lot was immersed in quality tested tap water (To) and other three lots were dipped in 2% CaCl2 solution for 5 min prior to storage in modified atmospheric chambers under different concentrations of CO2. Physicochemical analyses of stored samples were then conducted and overall results designated that T3 (10% CO2 + 2% CaCl2 pre-storage dipping) effectively reduced gray mold incidence and well maintained other quality attributes followed by T2 (5% CO2 + 2% CaCl2) as second best treatment. T1(3% CO2 + 2% CaCl2) didn’t show any significant effect on quality traits while control grapes (0% CO2) started decaying at 4th day of storage and were completely spoiled at 8th day. Storage period comprised of 12 days and postharvest quality analyses were conducted with an interval of 3 days.
Keywords
Botrytis cinerea; Modified atmosphere; Synthetic fungicides; Total Phenolic Compounds (TPC); Total Viable Count (TVC)
Introduction
Grapes (Vitis vinifera L.) are grown worldwide as world’s 2nd most grown fruit crop due to their enhanced pharmaceutical, nutraceutical and economical value, belong to family Vitaceae (Queen of fruits). Botanically non-climacteric in nature, growing on perennial and deciduous woody wines, mainly consumed in wine making (71%), fresh fruit (27%) and as (2%) dried fruit [1]. In a developing country like Pakistan that has poor infrastructure and lack of postharvest technological skills, a significant segment of this delicate fruit is lost [2]. When harvested, stem dryness causes severe water loss, and this loss of water leads towards browning of grapes caused by molds particularly notorious gray mold. It is roughly estimated that flaws and inadequate facilities in postharvest handling, transportation, storage and marketing cause 16% to 23% loss of grapes in Pakistan [3] with an average of 20.5% of total crop, this needs to be prevented as an immediate approach to minimize these losses.
Several factors like climatic conditions, soil conditions, harvesting period, origin of the cultivar, sanitary and phytosanitary conditions, cultural techniques and degree of maturity are involved in maintaining grapes keeping quality [4]. It is a simple fact that the postharvest quality of fresh produce cannot be improved however; it can be maintained at a certain level.
Ripening of grapes correspond to a series of biochemical, physiological and structural factors such as color changes, firmness, volatile compounds and accumulation of soluble solid contents and sugars, organic acids oxidation and decreased alkaloids. Color and firmness are considered to be the most important attributes that determine the quality of the fruit. Apart from color and firmness, rachis browning caused by gray mold growth appears to be a major obstacle in novel storage methodologies for grapes because alternative developed technologies for storage of grapes do not have the aptitude to decrease the browning of the stems and berries [5].
To cope with this problem, various postharvest treatments have been proven effectively in delaying the postharvest physiology of fruit tissues to prevent it from fungal decay. Modified atmospheric storage conditions either alone or in combination with various other postharvest treatments can effectively be used to control the postharvest ripening of grapes by efficiently maintaining their end quality [6]. This storage doesn’t only keep the product safe but also meet the standards of safety [7]. Modified atmosphere with increased CO2 level and reduced O2 concentration either delayed or postponed the injury signs, microbial growth was also noticeably reduced at high concentrations of CO2 [8].
Modified atmosphere has various beneficial effects in terms of minimizing water loss, reduced metabolic activity, delayed stem browning, maximum color retention, reduced respiration activity, lowering microbial load, and minimal chilling injury among several other benefits [9]. The important thing to notice in modified atmospheric storage of fresh produce is selective nature of storage product, CO2 t o O2 permeability and the packaging material. Highly reliable gas in fruits storage is considered to be CO2 that is both water and lipid soluble having bacteriostatic assets as well. Thus, the overall effect of CO2 on microbial load is the addition of lag phase of growth and decreased size during logarithmic phase. However, the latter consequence is greater as bacteria move from lag to log phase of evolution during which inhibitory effects are almost condensed [10]. Controlled and modified atmosphere technologies have significant effects on rachis browning of stems and making the product safe for intended use by successfully extending their shelf life [5].
Several authors have stated that modified atmospheric storage of fruits is becoming a prevalent method of prolonging the shelf life, but escalation in storage period also increases the probability of mold and bacterial decomposition of the product. The atmosphere inside the packages may not be as low in O2 or high to an adequate amount in CO2 to prevent the multiplying of spoilage entities [11]. TSS, TA and TSS/TA ratio are the chief quality factors defining the grapes acceptability by its consumers [12] and these are the same parameters that are drastically affected during storage.
Apart from this modified storage, different salt pre-treatments can also play a vital role in maintaining the quality attributes of the fruits. Calcium is regarded as playing a key role in establishing the cell wall structure of fruits by noticeably interacting with pectic acid to generate calcium pectate by initiating the cross linkages of pectic polymers. Calcium chloride (CaCl2) that is a salt by nature has extensively been used in fruits and vegetables industry as a preservative and firming tool, generally recognized as an ionic compound of chlorine and calcium, behaving as typical ionic halide. It also has been shortlisted as a permitting food additive with E-number of E509, also considered as safe (GRAS) by the U.S. Food and Drug Administration.
The study in hand is in continuation to previous study where level of CO2 at different storage chambers was fixed at 5% and different concentrations of CaCl2 were used, out of which 2% CaCl2 treated grapes showed significant results, keeping in mind, in present study effect of different concentrations of CO2 was analyzed by fixing CaCl2 level at 2%.
Materials and Methods
Freshly harvested grapes were procured from an orchard of Faisalabad city, Punjab, Pakistan. The berries were then brought to Food Safety Laboratory of National Institute of Food Science and Technology, University of Agriculture, Faisalabad for further processing and storage.
After sorting and grading, healthy clusters were divided in 4 equal lots. 1st lot was simply immersed in tap water and was entitled as control sample (T0), other three lots after being dipped in 2% CaCl2 solution were stored at 3% CO2 (T1), 5% CO2 (T2) and 10% CO2 (T3) modified atmospheric storage chambers (Memmert ICH260, Germany).
This dipping pre-treatment to all samples at ambient conditions was given for 5 minutes followed by packing in polypropylene perforated (2%) storage bags and stored at different chambers. In all four storage chambers, temperature and relative humidity were attuned at 10 ± 1°C and 80% as shown in treatment plan (Table 1).
| Treatments | CO2 Level (%) | CaCl2 Conc (%) |
|---|---|---|
| T0 | 0 | 0 |
| T1 | 3 | 2 |
| T2 | 5 | 2 |
| T3 | 10 | 2 |
Table 1 Treatment plan.
Total sugars and TSS
Total sugars were determined in terms of g/100 g by following the procedure (Method No. 967.21) as described in AOAC [13] and total soluble solids (°Brix) were measured using digital hand Refractometer (Carl Zeiss Jena-Germany).
Titratable acidity (%)
TA of grapes was determined by titrating the diluted sample against 0.1 N NaOH with phenolphthalein indicator according to method described by AOAC [13].
Firmness (N/mm)
Firmness of the berries was measured by using texture measuring system fitted with needle probe. Berries were randomly selected from each treatment and placed at the base of the texture analyzer (Mod. TA-XT2, Surrey, UK). The force (N) required to penetrate the fruit surface up to a specific depth (mm) was recorded and expressed in terms of N/mm [14].
Total polyphenols
After preparing the sample, total phenolic compounds were estimated by Folin-Ciocalteu method [15] with slight modifications by using an ultraviolet visible spectrophotometer (CECIL CE7200, UK). The absorbance of the samples was recorded from UV-Vis spectrophotometer at 275nm. Gallic acid was run as a standard along with the samples and its standard curve was used for the calculation of the total phenolic contents in the samples.
Total viable count (log CFU/g)
Berries of known weight from treatments were washed in sterile distilled water using a shaker, with fruit to distilled water ratio as 1:9. The wash water was then further diluted using peptone water, up to 105 and plated in triplicate on potato dextrose agar prepared as per manufacturer’s instructions and incubated at 25°C for 24 hours to count the microbial colonies [16].
Statistical analysis
The data was scrutinized statistically to determine the level of significance [17]. Experiment was a completely randomized design with factorial arrangement. Difference and comparison between the means were evaluated by LSD multiple range test at 5% level of significance.
Results and Discussion
Important challenge in postharvest shelf life extension of grapes is the effective control of notorious gray mold that has direct link with other biochemical quality attributes, so this was achieved successfully in present study and quality parameters were well maintained at acceptable level. T3 (10% CO2 + 2% CaCl2) was the best treatment among all mentioned quality parameters with statistically highly significant results and T0 as the worst followed by T1, however T2 showed fair results.
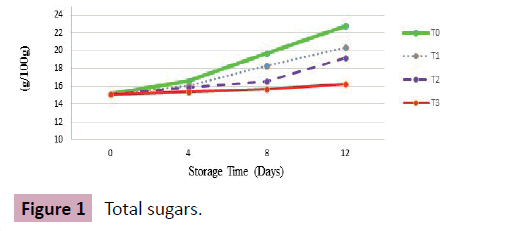
Total sugars (g/100 g)
Results: A significant portion of soluble solids in grapes is of sugars among which fructose and glucose are abundant while sucrose is non-reducing with level not exceeding beyond 1%. In present study, a continuous increment in soluble solids content and sugars was noticed as depicted in (Table 2). Grapes at harvest showed mean value of 15.14 g/100 g of total sugars that showed an increasing trend with increase in storage days.
| Storage Days | Treatments | Total Sugars (g/100g) | TSS (° Brix) | TPC (mg of GAE 100g-1 | TVC (log CFU g1) | Firmness (N/mm) | TA (%) |
|---|---|---|---|---|---|---|---|
| 0 | -- | 15.14 ± 0.4 | 16.6 ± 0.7 | 172.1 ± 5.7 | 2.68 ± 0.1 | 2.81 ± 0.09 | 0.84 ± 0.07 |
| 4 | T0 | 16.59 ± 0.6 | 21.3 ± 1.3 | 154.2 ± 5.3 | 3.2 ± 0.2 | 2.72 ± 0.3 | 0.72 ± 0.07 |
| T1 | 16.1 ± 0.2 | 18.1 ± 0.8 | 161.7 ± 5.4 | 2.59 ± 0.3 | 2.75 ± 0.2 | 0.77 ± 0.05 | |
| T2 | 15.9 ± 0.2 | 17.2 ± 0.8 | 166.4 ± 5.8 | 2.53 ± 0.4 | 2.73 ± 0.1 | 0.8 ± 0.06 | |
| T3 | 15.4 ± 0.5 | 16.9 ± 1.0 | 169.2 ± 9.5 | 2.46 ± 0.09 | 2.79 ± 0.1 | 0.83 ± 0.05 | |
| 8 | T0 | 19.7 ± 0.9 | 25.9 ± 2.0 | 114.6 ± 4.3 | 3.8 ± 0.1 | 2.45 ± 0.13 | 0.69 ± 0.03 |
| T1 | 18.3 ± 0.3 | 19.2 ± 1.0 | 129.6 ± 5.1 | 2.68 ± 0.2 | 2.56 ± 0.1 | 0.71 ± 0.04 | |
| T2 | 16.6 ± 0.8 | 18.8 ± 1.0 | 141.8 ± 5.8 | 2.55 ± 0.1 | 2.64 ± 0.2 | 0.76 ± 0.09 | |
| T3 | 15.7 ± 0.3 | 17.3 ± 1.0 | 162.9 ± 6.3 | 2.51 ± 0.09 | 2.75 ± 0.2 | 0.81± 0.04 | |
| 12 | T0 | 22.8 ± 1.6 | 29.7 ± 2.9 | 78.5 ± 4.5 | 4.22 ± 0.3 | 1.9 ± 0.08 | 0.61 ± 0.06 |
| T1 | 20.4 ± 0.2 | 25.4 ± 2.1 | 104.1 ± 6.8 | 2.88 ± 0.1 | 2.1 ± 0.09 | 0.65 ± 0.07 | |
| T2 | 19.2 ± 0.2 | 23.6 ± 2.1 | 123.2 ± 5.2 | 2.82 ± 0.2 | 2.53 ± 0.1 | 0.71 ± 0.04 | |
| T3 | 16.3 ± 0.4 | 18.1 ± 0.5 | 153.7 ± 5.7 | 2.54 ± 0.08 | 2.71 ± 0.1 | 0.79 ± 0.05 |
Given data are the mean values with 3 replications ± SD.
T0= Control (Water washed sample stored at 0% CO2 chamber), T1= 3% CO2 (2% CaCl2), T2=5% CO2 (2% CaCl2), T3=10% CO2
(2% CaCl2). Temperature (10 ± 1°C) & R.H (85%) was kept same in all 4 storage chambers.
Table 2 Effect of different concentrations of CO2 on physicochemical traits of grapes during storage.
Among all treatments, storage sample kept at 10% CO2 storage chamber after being dipped in 2% CaCl2 solution was T3 that showed statistically significant results with relatively un-noticeable increase in sugar contents that reached 15.4, 15.7 and 16.3 (g/100 g) at 4th, 8th and at 12th day of storage respectively. Similarly, grapes stored at 5% CO2 chamber (T2) was the second-best treatment with average mean value of 19.2 at 12th day of storage followed by T1 (3% CO2 storage chamber). An uncontrollable increase in sugar contents was observed in T0 (0% CO2) where from harvest (15.14) to termination day, a harsh increase in sugar contents was recorded with mean values of 16.59, 19.7 and 22.8 at 4th, 8th and at 12th day of storage as shown (Figure 1).
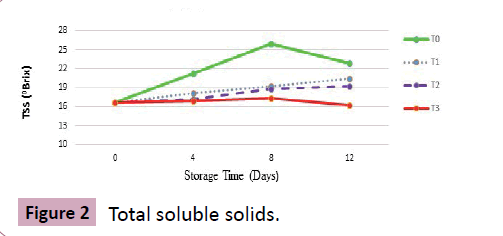
Total soluble solids (°Brix)
Results: About soluble solids, the value at harvest was recorded as 16.6° (Brix) that also showed an inclined trend towards storage days irrespective of the given treatment and storage conditions.
T0 showed rapid quality degradation with mean values of TSS as 21.3°, 25.9° and 29.7° at 4th, 8th and at 12th day of storage. T1 showed mean value of 25.4° with slight difference from T2 that showed average value of 23.6° at 12th day of storage. Highly significant effect of treatments was observed in T3 where starting from harvest to termination, only slight increment in TSS was recorded proving the treatment most effective with mean values of 16.9°, 17.3° and 18.1° at the day of termination as shown (Figure 2).
Discussion: This increase in TSS and total sugars is attributed towards breakdown of sugars and their conversion into starch and insoluble carbohydrates. An interesting thing noticed was higher concentration of CO2 in combination with post-harvest CaCl2 dipping formed a layer around the surface of the fruit that actively delayed the ripening process. This increase in TSS and total sugars was more rapid after 8 days of storage that was possibly due to hydrolysis of different polysaccharides. These findings are in close resemblance with the results of Sabir et al. [18]. Furthermore, findings of Javed et al. [19] also endorses the current study illustrating that with increase in CO2 concentration during modified atmosphere storage, slower increase in sugar contents was recorded.
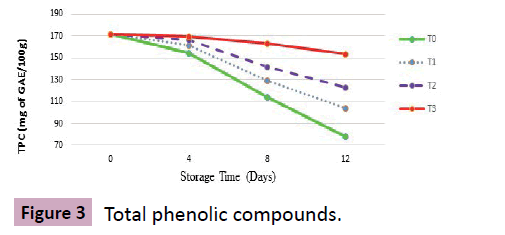
Total phenolic compounds (mg of GAE/100 g)
Results: It is evident from mean squares regarding total polyphenols (TPC) of treated grapes that significant variations were recorded against the effect of treatments and storage days. At harvest, berries showed mean square value of 172.1 mg of GAE/100 g of total phenolic compounds that were continuously decreasing irrespective of the given treatment with advance in storage days as shown (Figure 3).
Among all treatments, T3 showed statistically significant results with slight decrease in TPC that reached 169.2, 162.9 and 153.7 (mg of GAE/100 g) at 4th, 8th and at 12th day of storage respectively. Similarly, T2 with average square mean value of 123.2 at 12th day of storage. A noticeable fall in TPC was observed in T0 where from harvest (172.1) to termination day, a rapid decrease in TPC was recorded with mean square values of 154.2, 114.6 and 78.5 at 4th, 8th and at 12th day of storage as shown (Table 2).
Discussion: Among all fruits and vegetables, highest total polyphenols are present in grapes [20] however, wine grapes higher concentration of TPC than table grapes [15]. Findings of present study are in confirmation with the findings of [21] who reported that the anthocyanin and polyphenols of grapes were decreasing during storage period irrespective of the treatment provided prior to storage.
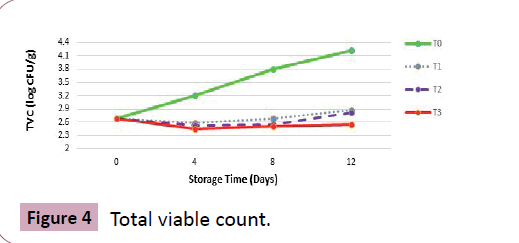
Total viable count (log CFU g-1)
Results: Findings in (Table 2) reveal that at harvest, the total viable count (TVC) was recorded as 2.68 log CFU g-1 that decreased in all treatments after being dipped in 2% CaCl2 solution apart from T0 that tend to increase throughout storage indicating no effect of normal tap water washing in controlling the microbial load.
Maximum incidence of browning and fungal decay caused by Botrytis cinerea was recorded in T0 that increased extravagantly at the later part of storage. In T0, after harvest the level of TVC was recorded as 3.2, 3.8 and 4.22 at 4th, 8th and at 12th day of storage that indicates maximum growth of gray mold development in control sample depicting maximum quality decay. However, the incidences of microbial load were recorded as 2.82 in T2 and 2.88 in T1 at the storage termination day.
Maximum quality preservation and reduced gray mold development was recorded in T3 that has higher CO2 concentration which made it possible to effectively reduce the presence of fungal pathogens. Starting from harvest (2.68), a statistical significant decrease in microbial load was recorded as 2.46, 2.51 and 2.54 that were even lesser that at harvest proving the treatment as most effective (Figure 4).
Discussion: The typical practice to minimize postharvest decay of berries globally is fumigation with sulfur dioxide (SO2). Fumigation of the fruit after harvest with SO2 gas effectively controls the decay but severe health issues of its residues have been widely reported. Among several fungal pathogens that are responsible for postharvest quality decay of fruits, gray mold top the list with highest rate of cases reported [22].
Investigations of present study are in close resemblance with findings of Chen et al. [23] who reported that high CO2 atmosphere is effective in controlling the development of B. cinerea in berries during prolonged storage. This declining microbial load trend in table grapes during storage is also in corroboration with the findings of Guillen et al. [24] who studied the overall quality characteristics of table grapes under different modified atmospheric storage conditions. A deadly attack of Botrytis c. in healthy bunch of grapes at the time of harvest is shown (Figure 5).
Firmness (N/mm)
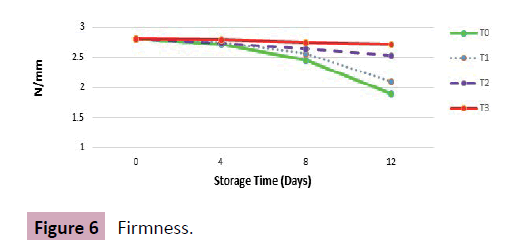
Results: Freshly procured grapes at harvest showed mean square firmness of 2.81N/mm that decreased with prolonged storage varying upon storage conditions and given treatments.
All treatments, T0, T1, T2 and T3 showed mean square values of 1.9, 2.1, 2.53 and 2.71 at storage termination day. Greater the force required by the probe to puncture the berries, more firm the berries are. Amongst all treatments, best firmness readings were recorded in T3 as 2.79, 2.75, and 2.71 closely followed by T2 with 2.75, 2.55 and 2.53 at 4th, 8th and at 12th day of storage respectively. T0 depicted lowest mean square values of 2.72, 2.45 and 1.9 at last day of storage indicating complete loss of berries firmness and amassing of cell structure (Figure 6).
Discussion: Possible reason behind degradation of berries skin is rapid loss of water contents and increased incidences of fungal pathogens. As storage period advances, the force required for the probe of texture analyzer to penetrate the berries also decreases. Our findings of decreased berry firmness are also in accordance with the findings of Martinez-Romero et al. [11], who reported that modified atmosphere packaging maintains quality of table grapes, and showed that at harvest, table grapes showed firmness of 2.75 ± 0.16 N/mm that decreased as the storage period prolonged. At termination day of storage, minimum loss in firmness was noted in modified atmosphere packages as compared to control indicating lower the O2 and greater the CO2 inside the chamber, higher the value of firmness was recorded.
Titratable acidity (%)
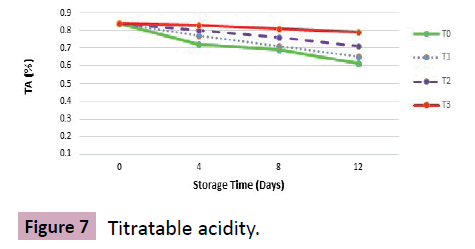
Results: Titratable acidity (TA) of grapes under different treatments and storage conditions regarding mean square, significant variations were recorded. TA was decreasing as storage period was advancing. Amongst treatments, a similar behavior was shown by all treatments indicating a steady decrease in TA value during storage (Figures 7-10).
Grapes at harvest showed a TA value of 0.84% that decreased with prolonged storage varying upon storage conditions and given treatments. The mean value of the treatment under which stored sample was kept at 0% CO2 (T0) depicted significant decrease in TA from 0.84 at harvest to 0.72, 0.69 and 0.61 at 4th, 8th and at 12th day of storage respectively followed by T1 with mean square value of 0.65 at storage termination. Results of T2 and T3 were in closed resemblance succeeded by T3 with average values of 0.83 at 4th, 0.81 at 8th and 0.79 at 12th day of storage.
Discussion: Consumer preference of table grapes mainly depends on taste and this delicious taste relies on TA that was decreasing throughout. This fall in TA is probably due to the depletion of organic acids and their conversion into sugars. Organic acids represent 0.5% to 1.5% of the berry weight, with tartaric acid being predominant with respect to malic and citric acids [25]. Acidity was also reduced because malic enzyme was increased in fruit during postharvest ripening and maturation. Another study suggested that acidity was reduced due to pyruvate decarboxylation reaction in which metabolic activity of living tissues significantly decreased. Overall judgments of the current study are in accordance with the evaluations of Randhawa et al. [26] and with the findings of Lin et al. [27] who investigated the effect of high CO2 concentrations on finished product quality of button mushrooms under modified atmosphere storage.
Conclusion
The Findings of present study show that different concentrations of CO2 inside the modified atmospheric storage chambers have direct correlation with biochemical characters of grapes. CaCl2 pre-storage dipping in combination with modified atmosphere storage showed a positive impact in preserving an attractive and healthy nutritional profile. However, among all treatments, grapes stored at 10% CO2 storage chamber (T3) has better quality profile and were well acceptable by the judges at storage termination day while grapes stored at 5% CO2 storage chamber were also acceptable up to 8th day. Control grapes (T0) showed rapid decaying process even at 4th day and were completely spoiled at 8th day of storage. Overall, physicochemical attributes of grapes after being dipped in 2% CaCl2 solution and stored at 10% CO2 storage chamber can effectively and efficiently preserve the quality of this delicate fruit.
Conflicts of Interest
The authors don’t have any conflict of interest.
Acknowledgement
The authors thank Supervisors and Director General National Institute of Food Science and Technology, University of Agriculture, Faisalabad for their kind support and encouragement during the conduction and completion of this work.
References
- Yang Q, Wang H, Zhang H, Zhang X, Apaliya MT (2017) Effect of Yarrowia lipolytica on postharvest decay of grapes caused by Talaromyces rugulosus and the protein expression profile of T. rugulosus. Posthar Biol Technol 126: 15-22.
- GOP (2009) Agriculture statistics of Pakistan, 2008-09. Ministry of Food, Agriculture and Livestock. Economic Wing, Islamabad, Pakistan.
- Nisar AS, Khan S, Kasi MA, Khair SM (2002) Postharvest and cold storage losses of apples in Baluchistan. Asian J. Plant Sci 1: 65-66.
- Khair SM, Ahmad M, Ehsanullah I (2009) Profitability analysis of grape orchards in ‘Pishin’ Baluchistan: An ex-post analysis. Sarhad J. Agricul 25: 103-111.
- Lichter A (2016) Rachis browning in table grapes. Austral. J. Grape Wine Resear 22: 161-168.
- Valero D, Valverde JM, Martinez-Romero D, Guillen F, Castillo S, et al. (2006) The combination of modified atmosphere packaging with eugenol or thymol to maintain quality, safety and functional properties of table grapes. Posthar Biol Technol 41: 317-327.
- Ortega-Regules A, Ros-García JM, Bautista-Ortín AB, López-Roca JM, Gómez-Plaza E (2008) Differences in morphology and composition of skin and pulp cell walls from Grapes: Technological Implications. Euro Food Res Technol 227: 223-231.
- Ahmad W, Junaid M, Nafees M, Farooq M, Saleem B (2004) Effects of pruning severity on growth behavior of spur and bunch morphology of grapes. Int. J. Agricul. Biol 6: 160-161.
- Deng Y, Wu Y, Li Y (2006) Physiological responses and quality attributes of ‘Kyoho’ grapes under Control Atmospheric Storage. Food Sci. Technol 39: 584-590.
- Singh P, Wani A, Saengerlaub S, Langowski HC (2011) Understanding critical factors for the quality and shelf-life of MAP fresh meat: a review. Crit. Rev. Food Sci. Nutri 51: 146-177.
- Martinez-Romero D, Guillen F, Castillo S, Valero D, Serrano M (2003) Modified atmosphere packaging maintains quality of table grapes. J. Food Sci 68: 1838-1843.
- Crisosto CH, Palou L, Garner D, Armson DA (2002) Concentration by time product and gas penetration after marine container fumigation of table grapes with reduced doses of sulfur dioxide. Hort. Technol 12: 241-245.
- AOAC (2006) Official methods of analysis. The Association of Analysts. Chem. (18th edn). Gaithersburg, Maryland, USA.
- Tian SP, Jiang AL, Xu Y, Wang YS (2004) Responses of physiology and quality of sweet cherry fruit to different atmospheres in storage. Food Chem 87: 43-49.
- Marinova D, Ribarova F, Atanassova MA (2005) Total phenolics and total flavonoids in Bulgarian fruits and vegetables. J Uni Chem Tech Metallur 40: 255-260.
- Badosa E, Trias R, Pares D, Pla M, Montesinos E (2008) Microbiological quality of fresh fruit and vegetable products in Catalonia using normalized plate-counting methods and real time polymerase chain reaction. J Food Sci. Agricul 88: 605-611.
- Montgomery DC (2008) Design and analysis of experiments. John Wiley & Sons.
- Sabir FK, Sabir A, Kara Z (2010) Effects of modified atmosphere packaging and ethanol treatments on quality of minimally processed table grapes during cold storage. Bulgarian J Agricul Sci 16: 678-686.
- Javed MS, Randhawa MA, Butt MS, Nawaz H (2015) Effect of calcium lactate and modified atmosphere storage on biochemical characteristics of guava fruit. J. Food Process Preserv 40: 657-666.
- Ghouila Z, Laurent S, Boutry S, Vander Elst L, Muller RN, et al. (2017) Antioxidant, antibacterial and cell toxicity effects of polyphenols from ‘Ahmeur Bouamer’ grape seed extracts. J. Fundament. Appl. Sci 9: 0392-410.
- Pinheiro ES, Costa JMC, Clemente E (2009) Total phenolics and total anthocyanin found in grapes from ‘Benitaka’ cultivar. J. Food Technol 7: 78-83.
- Romanazzi G, Feliziani E, Baños SB, Sivakumar D (2017) Shelf life extension of fresh fruits and vegetables by chitosan treatment. Critic. Rev. Food Sci. Nutri 57: 579-601.
- Chen SJ, Zhang M, Wang SJ (2011) Effect of initial hermetic sealing on quality of ‘Kyoho’ grapes during storage. Posthar Biol Technol 59: 194-199.
- Guillen F, Zapata PJ, MartínezÃÆâÃâââ¬ÃâÃÂRomero D, Serrano M, Valero D (2007) Improvement of the overall quality of table grapes stored under modified atmosphere packaging in combination with natural antimicrobial compounds. J. Food Sci 72: 185-190.
- Ali S, Khan AS, Malik AU, Shahid M (2016) Effect of controlled atmosphere storage on pericarp browning, bioactive compounds and antioxidant enzymes of litchi fruits. Food Chem 206: 18-29.
- Randhawa MA, Rashid A, Saeed M, Javed MS, Khan AA, et al. (2014) Characterization of organic acids in juices of some Pakistani citrus species and their retention during refrigerated storage. J. Anim. Plant Sci 24: 211-215.
- Lin Q, Lu Y, Zhang J, Liu W, Guan W, et al. (2017) Effects of high CO2 in-package treatment on flavor, quality and antioxidant activity of button mushrooms during post-harvest storage. Posthar. Biol. Technol 123: 112-118.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences